New group paper: Halogen behaviour in subduction zones: eclogite facies rocks from the Western and Central Alps
After exactly 53 weeks of writing, revising, collecting new data and riding the emotional rollercoaster that is peer-reviewed publication, my first paper as lead author was accepted for publication in Geochimica et Cosmochimica Acta. Having invested so much time and effort into that piece of work, I’m not sure whether my strongest feeling was of happiness or relief after the email confirming acceptance came through (at 5 am on a Saturday!).
The paper is about the behaviour of halogens — fluorine, chlorine, bromine and iodine — during the process of subduction of oceanic crust. Halogens are a highly volatile and reactive family of elements. Volatile elements such as these can affect certain physical properties within the interior of Earth (e.g. its rheology) and may influence the style and pattern of convection in the mantle and, therefore, heat transfer to the surface, which governs the life span of a planet. Outside the interior of Earth, halogens are also thought to be efficient at destroying the ozone layer, so the more we understand about their behaviour, the better!
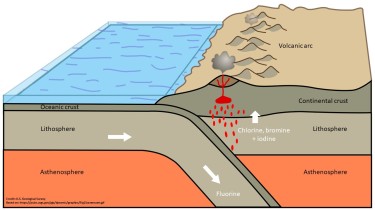
A typical convergent plate margin showing an oceanic plate being subducted below a continental plate, with the associated volcanic arc. (Image courtesy of the U.S. Geological Survey)
Subduction occurs at the boundaries of tectonic plates and involves the sinking of one plate beneath the other due to differences in density, resulting in the recycling of crustal material back to the mantle. During the descent of the plate, various chemical changes happen to the rocks and minerals that make up the crust as pressure and temperature increases and they transform into more dense minerals. Some of these minerals contain water and other volatile elements such as the halogens in their structure, and as they break down into denser minerals, this water is released, and often takes the volatiles with it. Many of the minerals that contain water break down at low temperatures and pressures, such that water and volatiles are often lost quite early during subduction. However, some minerals that contain water can withstand surprisingly high temperatures and pressures, and it is these high temperature, high pressure “hydrous” minerals that are of particular interest to scientists. Water and volatiles have much lower densities than their surroundings, therefore, as they escape from the subducting crust, they rise into the tectonic plate above. This influx of water and volatiles to the dry crust above causes the minerals and rocks to begin melting and initiates a chain of volcanoes along the length of the plate boundary. This can be seen today in volcanic mountain ranges in regions such as the Andes and Japan.
The question is, how much water or volatile elements such as the halogens remain in the sinking crust that are able to be delivered back into the mantle, after most of the hydrous minerals have broken down? One of the problems we face is that we cannot access the rocks in subduction zones to measure their volatile content, because they are too deep to see or interact with. Luckily, nature provides us with an alternative way. Once the process of subduction is complete upon the closure of an ocean, the sinking oceanic crust breaks off from the tectonic plate, leaving a small section behind — an ophiolite — which rises and becomes exposed at the surface as continents collide to form mountain belts such as the Alps and the Himalayas of today. We can then easily go to collect these rocks and bring them to the lab for analysis.

Halogens are converted to noble gases by neutron irradiation, and then the Thermo Argus IV noble gas mass spectrometer is used to analyse the noble gases (Image credit: Isotope Group)
In this study we use sophisticated techniques called electron-probe micro-analysis (EPMA), time-of-flight secondary ion mass spectrometry (TOF-SIMS) and neutron-irradiated noble gas mass spectrometry (NI-NGMS) to measure the tiny concentrations of halogens within our high-pressure mineral samples. As it turns out, despite being part of the same periodic group, halogens like to behave quite differently during subduction. Fluorine likes to hang around with the mineral and prefers not to leave with the fluid generated during the breakdown of hydrous minerals, such that it may travel to great depths within the mantle. Chlorine, bromine and iodine instead prefer to leave the mineral phase and travel with the fluid. However, it seems that chlorine prefers to do this much earlier during the subduction process, at shallow depths, whereas it takes a little longer for the bromine and iodine to escape the subducting slab. However, it’s safe to say that after most of the hydrous minerals have broken down during subduction, little chlorine, bromine and iodine

A variety of sample rock types collected from various locations in the Alps. A property called birefringence causes rocks to look like stained-glass windows under the microscope. (Image: L. Hughes)
remains to be taken into the mantle. The behaviour of chlorine, bromine and iodine makes them great tracers for us to see how volatiles are transported around the different geochemical reservoirs of Earth. Fluorine, on the other hand, behaves quite differently to the rest of the halogen family, and is quite happy to stick around in remaining hydrous minerals of the sinking slab, as it helps them to remain stable under higher temperatures. Other studies have shown that deep within the mantle, in a region known as the transition zone, where the lower mantle meets the upper mantle, minerals exist that can accommodate significant amounts of water. These minerals, such as Wadsleyite and Ringwoodite, may collectively contain up to three times as much water as all the world’s oceans! And may, therefore, be a final storage site for fluorine, and perhaps other volatile elements too.
More work remains to be done on the behaviour and storage of volatile elements in the deep mantle, but studies such as these are helping us to understand the origin and evolution of planetary interiors, occurring hundreds of kilometres below our feet, from the comfort of a lab at the surface, and that is pretty cool!
Full citation: Hughes L., Burgess R., Chavrit D., Pawley A., Tartèse R., Droop G., Ballentine C and Lyon I.C. (2018) Halogen behaviour in subduction zones: eclogite facies rocks from the Western and Central Alps. Geochim. Cosmochim. Acta. 243, pp. 1-23.
DOI: https://doi.org/10.1016/j.gca.2018.09.024
url: https://www.sciencedirect.com/science/article/pii/S0016703718305490?via%3Dihub

